If you’re wondering how to participate in a clinical study, here are a few tips: Plan ahead, understand the risks and benefits, and sign the Informed Consent Form. In addition, you may want to ask about travel compensation if participating in a clinical study. Traveling can be expensive, so you’ll want to factor that into your budget. The researchers will advise you on how to make the travel arrangements.
Planning ahead
There are many logistics involved in a clinical study, and it is crucial to organize them ahead of time. Having all of your important documents in one place is essential, as it will make cross-checking easier. You should also consider planning how to communicate the trial with family and friends. If you can travel to your study, you should plan to reimburse yourself for travel expenses.
Based on vial.com, you should plan for everything in advance, from the site visits to the medication. The materials used for trial patients should be easy to read, and site staff should be trained to answer patient questions and concerns. Make sure the trial protocol is realistic, considering daily life and the symptoms of the disease. In some cases, patients will have difficulty opening packaging or following instructions. You should also consider the physical limitations of participants and develop trial protocols accordingly.
In most cases, clinical trials follow strict federal guidelines and safeguards to ensure the safety of participants. The clinical trial protocol is meticulously regulated and details the study’s procedures. The study results are reported to medical journals, scientific meetings, and various government agencies. However, the names and details of study participants are kept confidential. In addition, if you are selected to participate in a clinical trial, the process can be more complicated than you originally thought.
Understanding the risks and benefits of participating in a clinical trial
While participating in a clinical trial can come with some risks, it can also offer many benefits. For one, it can provide you with access to new treatments and improve the lives of many people. Furthermore, the trial results can help bring an effective medical solution to market, improving the lives of untold numbers of people. Finally, depending on the type of trial, you may receive free drugs or medical care as a result of your participation.
Considering participating in a clinical trial requires some research and preparation. Before consenting to participate, you must understand all the risks and benefits. Before signing up for a clinical trial, you must understand the information in the informed consent document and any additional information provided by the research team. Afterward, you should discuss your decision with your physician and other members of your family if you’re willing to help. Once you have read the consent document, send it back to the trial coordinator or investigator.
Although participation in a clinical trial involves risks, you will be helping people with kidney problems. You may receive a new treatment that is safer or more effective than the current standard of care. You may also receive free medical care since the study’s sponsors will cover your medical expenses. In addition, the study can help future generations suffering from kidney failure. In short, clinical trials can help us make a difference.
Informed consent process
The Informed Consent process is a document that gives the research subject a complete understanding of what they will be involved in, including the risks and benefits of the study. During this process, the participants should review the terms and conditions of the trial several times before signing it. The informed consent document should also include questions and possible injuries that may arise during the study. Once they have understood the information, they should sign the consent document and discuss it with others.
A study of 54 interventions reveals that most participants are willing to participate in the research. Informed consent is essential to avoid biases in clinical trials, so the authors recommend that participants complete the process to make informed decisions. It is not always possible to provide individuals with all the information they need to consent to a clinical trial. However, if the consent form does not contain the necessary information, the participants should not be excluded from the study.
A clinical trial is a meaningful way to test and develop new treatments. The process begins with a consent visit. The investigator, usually a physician, will explain the different options for treatment. Sometimes, only one investigative treatment is presented to patients, and they may feel forced to choose it. While this may sound like the ideal scenario, the participants are likely unaware of the full scope of the study and its risks. Moreover, the participants may not understand the full complexity of the trial, especially if it involves complex scientific reasoning. They may also experience information overload and difficulty concentrating in a stressful environment.
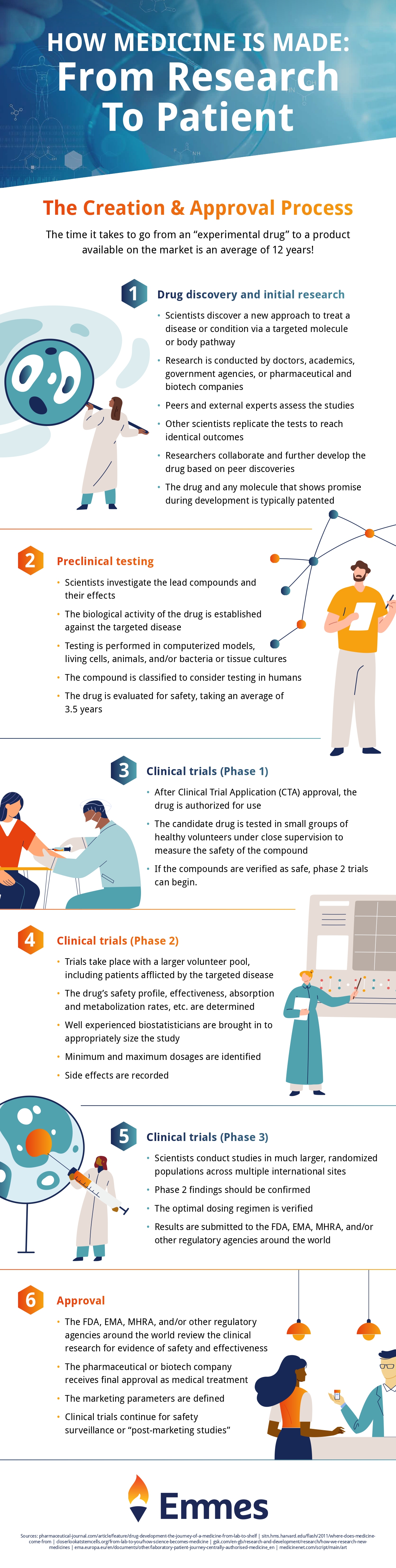
The journey to making medicine is a long and complex one, and clinical trials are just one step. Check out the infographic below to see the full story of how your medicine is made!
Infographic provided by The Emmes Company, a clinical data management organization